COVID-19 PCR Test (age 3+)
$89.00Sale!

FREE standard shipping
PCR Swab
Within 48 hours once the lab has your sample
Test in the comfort of your own home
Out of stock
Our COVID-19 Home Collection Kit DTC is a fast, affordable, and painless way to get tested for SARS-CoV-2, the virus that causes COVID-19. Detects the Delta variant and Omicron variant. FDA-authorized (EUA). Ages 3+.
Key information
Common Symptoms Cough, shortness of breath, fever
Measures SARS-CoV-2 virus (COVID-19) + Delta variant + Omicron variant
Collection Method Shallow, pain-free nasal swab
Tech RT-PCR
Results in 48 hours, Monday-Friday
Airline Approved for outbound international travel
For Ages 3 and up
Ship To The Lab Monday-Thursday only
$89.00Sale!
Out of stock
How it works

STEP 1
Order online
Get health tests delivered to your doorstep with just a few clicks
STEP 2
Collect at home
Complete your test right from your comfy couch, and mail it to our lab (Monday-Thursday).
STEP 3
Get secure results
Receive your test results in a few days & get the clarity you deserve, confidentially.
The details
Placing your order
Your order will be delivered within 2 business days. At home COVID tests are shipped Monday – Friday. If you order on Friday, the kit will arrive by the following Tuesday. Orders over the weekend will arrive by Wednesday. Make sure to order in advance to avoid possible shipping delays.
Collecting your sample
Returning your kit
Take your completed kit to a local FedEx drop box before end-of-day pickup to have it overnighted to the lab (Monday – Thursday only). FedEx drop boxes do NOT pick up on Saturday or Sunday. Check My Body Health is not responsible for FedEx shipping delays. Note: to avoid holiday shipping delays, please drop off your kit Monday – Thursday.
Results are ready
Your at home coronavirus test results will be available within 24-48 hours of the lab receiving your completed kit. You’ll get an email the moment your results are ready in our secure online portal. View, download and print them anytime. Feel free to share them with your healthcare provider.
Out of stock
Consider this test if you…
- Have mild cold-like symptoms
- Feel warmer than usual
- Have a fever above 100.4ºF
- Feel more tired than normal
- Have an elevated heart rate
- Have a dry cough or wet cough
- Plan to travel domestically or internationally
- Have shortness of breath
FAQs
Does this nasal PCR test have an age limit?
Yes! Our FDA authorization (EUA) only allows those at least 3 years of age to use the At Home COVID-19 Nasal PCR Test Kit.
What does it mean that this test is authorized by the FDA under emergency use authorization (EUA)?
Typically, FDA approval takes many years of testing and clinical trials. During a public health emergency (like the COVID-19 pandemic, the FDA can use its Emergency Use Authorization (EUA) authority to allow the use of unapproved medical products, or unapproved uses of approved medical products, to diagnose, treat, or prevent serious or life-threatening diseases when certain criteria are met, including that there are no adequate, approved, and available alternatives.
Can I be cleared for airline travel with this test?
Yes – this test is a molecular nasal PCR test, and it’s the test most airlines require to be cleared before you fly.
How do I meet the 72-hour time requirement for my airline?
-
Make sure to order your test well in advance of your upcoming flight, i.e. multiple weeks ahead of time.
-
Collect and return ship your sample to the lab 3 days before your flight. The “collection date” on your results will be the day/time you collected your sample and activated your kit. E.g. Collect on Monday afternoon for a Thursday morning flight. Make sure you drop off your completed kit before end-of-day pickup.
-
Your results will be ready within 24-48 hours after the lab receives your sample. This assumes no shipping delays. E.g. if you ship your kit to the lab on Monday afternoon before end-of-day pickup, your kit should arrive at the lab on Tuesday – results will be ready on Wednesday or Thursday. Note: Check My Body Health is NOT responsible for shipping delays.
-
We’ll email you when your results have posted in the empowerDX portal. Make sure to have them ready on your phone when you go to the airport. Download them and print the results as a backup.
Do you share my COVID-19 test results with anyone?
According to the CDC, “Yes, the CARES Act requires all clinical laboratories and testing providers that perform diagnostic testing under a Clinical Laboratory Improvement Amendments (CLIA) certificate to report the results of any test that is intended to detect SARS-CoV-2 or to diagnose a possible case of COVID-19 (e.g., molecular, antigen, antibody), not just positive tests, to state or local public health departments. Results from surveillance testing for SARS-CoV-2 should not be reported to state or local public health departments.”
But not to worry. Other than the CARES Act requirements above due to the pandemic, we keep your personal health information private. The Health Insurance Portability and Accountability Act (HIPAA) requires that your profile and results are only accessible to you and the Licensed Ordering Physician who reviews and approves your order.
Why does the “REF RANGE” section of my results say “Negative”?
A “Negative” “REF RANGE” has nothing to do with your actual test results.
“REF RANGE” means “reference range”. The reference range for vitamin D is 30-100, meaning that a normal or “healthy” level is between 30 and 100. The reference range for the At Home COVID-19 Nasal PCR Test Kit is “negative”, meaning that a normal result is “negative” – when NO SARS-CoV-2 virus is detected on the sample. Feel free to ignore this section.
Please refer to the “RESULT” section of your lab report to determine if your test results for SARS-CoV-2 are positive or negative. If the “RESULT” section of your report has a “NEGATIVE” under it, then your nose sample didn’t have the SARS-CoV-2 on it. If the “RESULT” section of your report has a “POSITIVE” under it, then the lab detected the SARS-CoV-2 virus on your nose sample.
Is it possible to get a false positive?
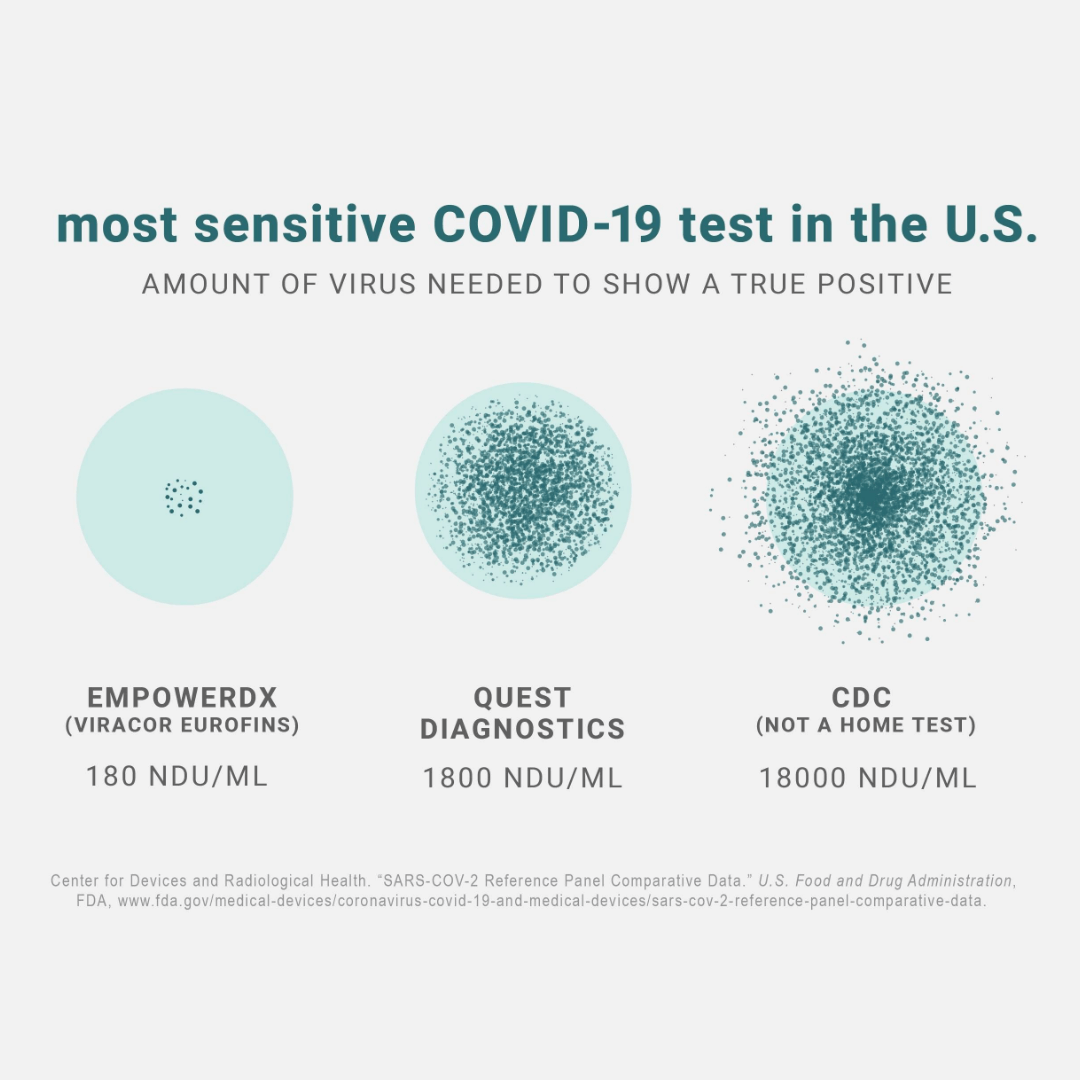
As of December 2020, our lab’s SARS-CoV-2 assay offers the best sensitivity of the 118 laboratories that have submitted results to FDA’s SARS-CoV-2 Reference Panel, with a limit of detection (LoD) of 180 NAAT Detectable Units/mL. We didn’t have any false positives during our FDA EUA validation study.
It is possible to have a positive test result and not be infected with COVID-19, but still have the SARS-CoV-2 virus in your nose.
If you have received a positive PCR test, we encourage you to follow CDC guidelines and have those around you get a PCR test as well.
Why do my results say TNP?
TNP means “test not performed”. The lab was unable to test your nasal sample for the SARS-CoV-2 virus due to an issue with your kit. Please contact Customer Care for next steps.
Why is the label on your website different than the label on the box I received?
We have received an updated EUA (emergency use authorization) from the FDA with new labeling required.
Will my result or shipment be delayed for holidays?
Yes. FedEx does not deliver on major holidays and our labs also close in observance of most major holidays. Note: to avoid holiday shipping delays, please drop off your kit Monday – Thursday.
Can this test detect the COVID-19 Delta variant and other variants?
Yes, the At Home COVID-19 Nasal PCR Test can detect all known variants of the COVID-19 virus, including the Alpha variant (B.1.1.7), Beta variant (B.1.351, B.1.351.2, B.1.351.3), Gamma variant (P.1, P.1.1, P.1.2), Delta variant (B.1.617.2, AY.1, AY.2, AY.3), Eta variant (B.1.525), Iota variant (B.1.526), Kappa variant (B.1.617.1), Lambda variant (C.37), and Omicron variant (B.1.1.529).
Should I tape my box shut before mailing?
No, there is no need to tape your kit box shut before returning it to the lab. Close your box and insert it into the Fedex Return pack. You will need to seal and mail back the Fedex Pack with return label attached.
Can I order more than one kit?
You are welcome to order more than one kit! Unfortunately, you will have to order each separately on our website as we do not allow users to order more than one in a single purchase. If you order a kit for someone else, the person who is collecting the sample must create a profile and match their name and date of birth on the collection tube.
Can I purchase a kit for someone else?
Absolutely. The person receiving the gifted kit must use the blank label within the kit. THIS IS NOT ALLOWED FOR COVID INSURANCE ORDERS. If you order through insurance, it must be used by the individual who placed the order.
How do I activate a kit for my family member or child?
A separate account is required for each family member activating a kit. The sample tube must have the name, date of birth, and date of collection of the family member or child using the kit. This name label must match the account profile when the kit is activated or results will NOT be offered.
Important information about my lab results
Lab results are intended for informational and educational purposes only, with the exception of infectious disease testing which is diagnostic. Information from empowerDX is not meant to serve as treatment advice or treatment recommendation for any health condition. We strongly recommend you review lab results with your physician.